
Add 0.15 ml glycerol (100%) to a 2 ml screw-cap vial and sterilize by autoclaving. Tip: Vials of sterilized glycerol can be prepared in batches and stored at room temperature until required. Add 0.85 ml of a logarithmic-phase E. coli culture to the vial of pre-sterilized glycerol.

During the study period, all solutions were observed to retain at least 95.26% of the initial concentration in both types of containers at both temperatures. Conclusions: Bortezomib (3.5 mg in manufacturer's vial) reconstituted with 1.4 mL NS is physically and chemically stable for up to 21 days at 4°C or 23°C when stored in either the

The selectivity of the method was assessed by the analysis of fruit and vegetable samples by the QuEChERS-USAE/UHPLC-PDA methodology and compared with a 10 μg/mL standard solution. The absence of interfering peaks at the elution times of the target analytes proves that the method is selective.

SureSTART Level 3 products are packed in a clean environment according to our own hygiene concept and regulations to protect against contaminating factors. The vials contain the lowest levels of extractable metals and leachables to ensure consistent, reproducible sensitivity and selectivity for the most challenging analytes.

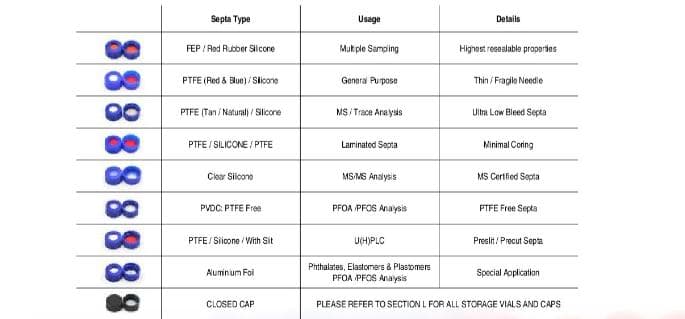
The vials contain the lowest levels of extractable metals and leachables to ensure consistent, reproducible sensitivity and selectivity for the most challenging analytes. The septa are conditioned to provide the lowest level of chromatographic extractables.

Headspace vials are the most popular vials for the analysis of gases and are suitable for headspace and SPME experiments. These products are suitable for GC systems. Sample storage Our 4, 8, 10, 16, 20 and 40 mL vials can be used for sample collection and sample storage. We have added a patch to these large vials, making it easy for you to
Apr 9, 2020 · The selectivity of the method was assessed by the analysis of fruit and vegetable samples by the QuEChERS-USAE/UHPLC-PDA methodology and compared with a 10 μg/mL standard solution. The absence of interfering peaks at the elution times of the target analytes proves that the method is selective.

Each 10 mg/mL, 2 mL vial contains methotrexate sodium equivalent to 20 mg methotrexate, and the following inactive ingredients: Sodium Chloride 0.70% w/v. Sodium Hydroxide and, if necessary, Hydrochloric Acid are added to adjust the pH to approximately 8.5. Each 25 mg/mL, 20 mL, 40 mL and 100 mL vial contains methotrexate sodium equivalent to 500

Algebra questions and answers. Nurses use proportions to determine the amount of drug to administer when the dose of the drug is measured in mg, but the drug is packaged in a diluted form in mL. For example, to find the number of milliliters of fluid needed to administer 300 mg of a drug that comes packaged as 120 mg in 2 mL of fluid, a nurse

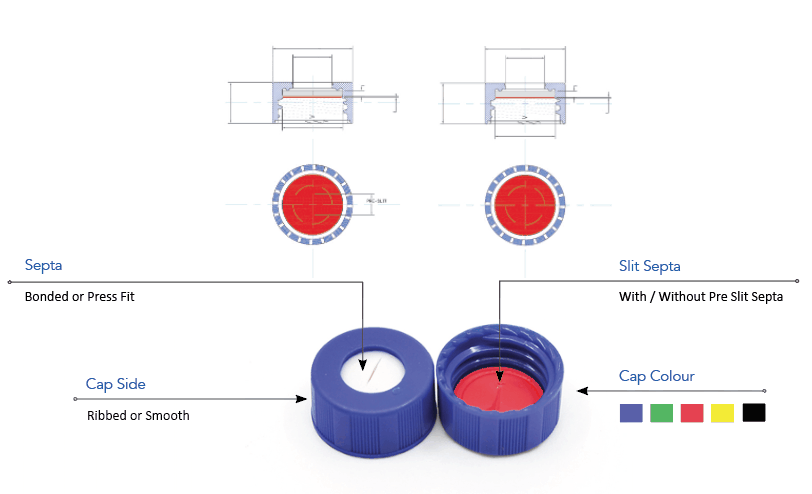
Vials, open-top screw cap, 2 mL large opening with closures, unassembled convenience packs 29118-U volume 2 mL, clear glass vial, thread for 10-425, black polypropylene cap, PTFE/silicone septa, pkg of × 100 ea

Sep 1, 2019 · A validation protocol for commercial sterility testing methods is proposed. •. Inclusivity determines the methods ability to detect the relevant microorganisms. •. LOD 95 determines the sensitivity of the selected methods. •. Performance of direct streaking and rapid commercial sterility methods are defined.

It is dispensed by your Pharmacist in small 2 mL vials which contain less than ½ teaspoonful of methotrexate. Here are some important features of the vials of methotrexate: ¾ For patients with inflammatory arthritis, methotrexate is supplied as 50 mg in a 2 mL vial (25 mg per 1 mL). ¾ The vial can be used for more than one dose because it
volume 2 mL, amber glass vial, O.D. × H × I.D. 12 mm × 32 mm × 4.6 mm, standard opening. View Pricing. Page 1 of 6. Find 2 ml glass vial and related products for scientific research at MilliporeSigma.

Syringe filters are single-use, membrane-based devices used for the removal of particulate impurities from liquid and gas samples prior to analysis by methods such as HPLC, ion chromatography, gas chromatography, ICP, and dissolution testing. Proper filtration of samples improves the quality and consistency of analytical results and decreases

For ionizable analytes, the results are very different retention profiles, and potential selectivity differences between the two fluorinated-phenyl columns. Figure 4. The effect of base particle on retention. Increased retention of famotidine (A) and famotidine tablet degradation products (B,C) on an HSS PFP stationary phase compared to early